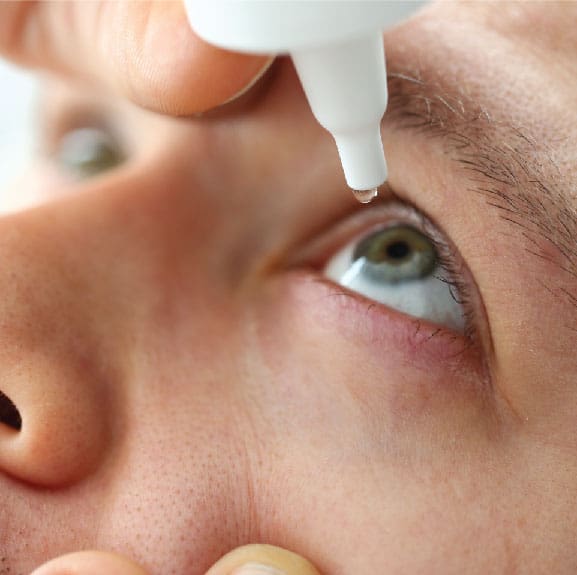
New FDA-approved eyedrops could improve close-up vision and replace reading glasses for millions of Americans.
A new eyedrop medicine, called Vuity, was approved by the FDA in October to treat presbyopia in adults. Presbyopia affects approximately 1.8 billion individuals worldwide and approximately 128 million Americans. Presbyopia is age-related and impacts the eyes abilities focus on close-up objects. It is a refractive error in the eye and occurs because the lens, the inner part of the eye that helps the eye focus, stops focusing on light correctly in the retina. 1 Presbyopia is part of the normal aging process and there is no way to stop or reverse presbyopia. However, it can be corrected through glasses, contact lenses, or eye surgery. 1 Now, presbyopia can be treated with eye drops (Vuity).
Vuity
What is Vuity and how does it work?
Vuity is an old medication used in a new way. The active drug in Vuity, pilocarpine, has been used to treat high pressure inside the eye caused by glaucoma and other eye diseases. However, Vuity is the first drug to utilize it to improve vision. Vuity works by using the eye’s natural ability to reduce pupil size. Reducing pupil size expands the depth of focus which allows a person to focus at different ranges naturally. By using once-daily drops of Vuity, patients have experienced enhanced close-up vision in as little as 15 minutes. One drop in each eye provides sharper vision for six to ten hours. The drops work best for ages 40-55 and are less effective after age 65. The medication is available by prescription only. A 30-day supply of the drug will cost about $80 and is not currently covered by insurance.
Two Phase III clinical trials led to FDA-approval of Vuity. Across GEMINI 1 and GEMINI 2 studies, there were 750 participants diagnosed with presbyopia aged 40 to 55 years old. Participants were randomized across both studies to be given either the placebo or Vuity. Patients treated with Vuity during the trials were able to read three or more lines on a reading chart 3 hours after receiving Vuity on day 30 of the trial without having their distance vision impacted in comparison to the placebo. No life threatening or serious adverse events were noticed in any of the participants.2
Vuity may be the only eyedrop for treatment of presbyopia on the market today, but competitors are coming up close behind. Eyenovia revealed that its drug device combination, MicroLine, is in the works for helping presbyopia patients. The product uses the company’s Optejet dispenser to deliver small doses of pilocarpine. The Optejet dispenser is a point of differentiation between Vuity drops and Eyenovia’s MicroLine.
Contact us if you have any questions about what pharmaceuticals can and should cover or what drugs better align with your prescription plans at https://mmaeast.com/contact/.
Sources:
- Kierstan Boyd. “What is Presbyopia?”, available at https://www.aao.org/eye-health/diseases/what-is-presbyopia, accessed 8 September 2022.
- Joseph M. Coney. “Vuity”, available at https://eyewiki.aao.org/Vuity, accessed 8 September 2022.